发布日期 2020年2月27日
孵化器的比较
摘要:
对4种常用的孵化系统用于鲑鳟鱼卵的孵化和卵黄吸收进行试验。这些系统包括上升流式孵化桶,和垂直式流水托盘孵化器的三种设置:敞开式、活动底托和固定底托。比较这些孵化系统的存活率、开口时鱼苗重量,以及平均重量到1克的时间。存活率在这些系统中没有明显差异。鱼苗在垂直式流水托盘孵化器的开放式设置中开口时重量最重,达到平均1克重量时间最短,开口重量和平均体重达1克的时间在垂直式流水托盘孵化器的所有设置中的结果,比上升流式孵化桶的结果平均要快14%。垂直式流水托盘的三种设置之间没有大的区别。
背景
商业化鳟鱼养殖场使用的孵化器有不同样式。基本上是,垂直式流水托盘(托盘垂直叠置)孵化器,和上升流式孵化桶。功能都是一致的,鱼卵发眼卵阶段放入孵化器进行孵化,直至卵黄苗的卵黄被吸收,但是,不同孵化器在孵化时的水压力和环境不同。这种环境的不同,给孵化后鱼苗造成的紧张度、能量消耗和整体健康也不同。一种孵化器是标准的上升流式孵化桶,有时称为麦克唐纳上升流孵化器。另外一种是立式托盘系统,通常称为叠置式托盘孵化器。在立式托盘系统里,我们选择了三种介质的底部形式。
方法
项目设计
本试验所用的鱼卵来自8月产卵种群的标准产品。受精和发眼培育是在位于华盛顿州,Sumner的设施内,按照鳟鱼庄标准操作进行的。在鱼卵的积温大约为210度日(ATUs)时,鱼卵经过剔除死卵和未发育的鱼卵,然后,装箱运至位于爱达华州双瀑镇岩石谷的基地。
鱼卵到达后,随机分成8个批次,每批次4000粒鱼卵。每4000粒鱼卵的批次放入以下述的4种不同的孵化器中。每种孵化器有一个重复对照。用32盎司的烧杯将鱼卵放入每个孵化器中。试验开始时,孵化器加入泉水(13.2℃),每个孵化器的水流量大约为每分钟6升。用黑色塑料将上升流式孵化桶包裹起来,使环境光线对鱼卵没有影响。
鱼卵在独立的系统中培育直至卵黄被吸收,之后移至不同的鱼苗槽中并开始投喂。
孵化系统
立式流水托盘孵化器
这是被广泛使用的鱼卵孵化系统,其设计是水流从上向下。典型的是根据水质和流量,4-8层托盘重叠放置。因为容易分别少数量设置,也可以在同一系统中孵化不同的鱼卵,因此,它们更常被州立机构用于研究目的使用。通常也被称为叠置式托盘孵化器。
立式流水托盘孵化器的底质
立式流水托盘孵化器有几种不同的底质。使用底质的原因是通过此介质,将托盘中的鱼卵分离开,减少真菌扩散,使新孵化出的幼苗有更少的能耗,改善托盘中的水流。底质是模仿溪流中自然小穴,给鱼的幼苗提供发育最适合的环境。理论上,增加鱼苗躲藏的地方,可使鱼苗更加强壮、健康和更少的能耗。
立式流水托盘孵化器使用:
- 敞开托盘式(C)
- 给鱼提供可躲过水流的地方
- 通过串联,水可以从上层托盘流到下层托盘
- 给鱼卵和鱼苗提供黑暗环境,开灯时光线也不能照入。
2 . 有可拆卸底质的托盘(DI)
- 与上面(敞开式)相同,只是增加了鱼躲藏水流的空间。
- 底质可以拆卸
3 . 有固定底质的托盘
- 与上两款相同,但底质是固定的。
孵化桶
孵化桶是由底部进入孵化器,流经发育中的鱼卵,从桶的上面流出。此系统的优点是,在卵黄被吸收后,鱼苗可以从孵化桶上面随水流入鱼苗培育槽内,不需要人工移出。然而,孵化桶中的水流搅动,使鱼苗游动消耗的能量而不是用于生长。
孵化桶的使用:
孵化桶(J)
- 上升流 – 提供了一个轻微搅动的环境
- 单一水流向
- 除非遮蔽环境光线,鱼苗在光照环境下。
Project Overview:

图1:整体项目设计 – 显示试验中不同的孵化系统


图2:不同底质
结果和讨论
结果和讨论
从试验结果我们可以得出结论,不同的孵化系统在存活率上没有明显差异。从发眼卵的孵化到孵化后鱼苗至1克,死亡率相近(表格1)。不同系统之间的差异小于1%,显示没有一种系统有明显优势。要注意的是,敞开式(C)系统与其他系统相比,更容易将死卵检出和维护。员工发现,可拆卸式底质(BI)和固定式底质(DI)使用不便。固定式底质(BI)难清洁,真菌会在底质上扩散,水流甚至不易流经托盘。达到孵化高峰期和到开口的时间,所有孵化器相似,在170度日(ATU)左右。孵化高峰期是指孵化达到70%的时候。.
| 试验组 | 鱼苗到1克时的存活率 (765.6 ATU) | Survival to 1g (765.6 ATU) |
| 存活率 % | 与试验组 C的差异 | |
| C | 88.7% | n/a |
| J | 88.4% | -0.3% |
| BI | 89.3% | 0.7% |
| DI | 89.2% | 0.6% |
表格1:不同孵化系统里鱼苗平均规格达到1克时的存活率。
重量
与敞开式孵化托盘相比,孵化桶里鱼苗开口时的体重规格要小15%(表格2)。我们相信这是鱼苗在孵化桶中抵抗水流,有更高的能耗,来保持向上游动的姿势。鱼苗体重规格与敞开式相比,固定式(BI)和可拆卸式(BI)的鱼苗分别轻2.6%和1.7%(表格2和图3),这三者间,在鱼苗耗能方面没有明显差异。
| 试验组 | 试验组 开口时 (171.6 ATU) | 开口时 (171.6 ATU) | 鱼苗1克时 (765.6 ATU) | 鱼苗1克时 (765.6 ATU) |
| 鱼苗体重 (g) | 与试验组 C的差异 | Weight (g) | 与试验组 C的差异 | |
| C | 0.115 | n/a | 1.02 | n/a |
| J | 0.097 | -15.7% | 0.88 | -13.7% |
| BI | 0.112 | -2.6% | 0.96 | -5.9% |
| DI | 0.113 | -1.7% | 1.01 | -1.0% |
表格2:从发眼卵到孵化高峰期的积温(ATU),以及从发眼卵到平均规格为1克时的积温(ATU)。
试验中观察到同样的趋势,敞开式系统中鱼苗平均规格达到1克时的积温为765 度日(ATUs)。此时,在敞开式系统中的鱼苗重量比孵化桶系统的要重13.7%(表格2和图3)。由此,我们可以得到的结论是,使用孵化桶孵化没有优势,要达到平均规格重量为1克时,需要更长的时间。
鱼苗在固定式底质系统和可拆卸式底质系统与敞开式底质系统相比,轻5.9%和1%,其结果再次显示敞开式系统没有明显优势(表格2和图3)。
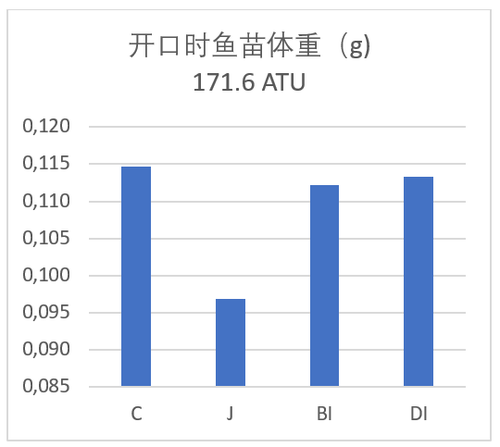
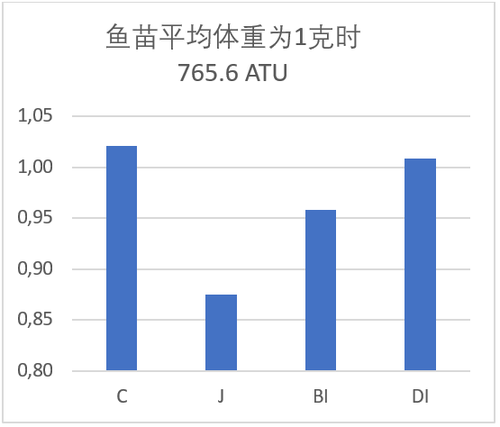
图3:图示幼苗开口时的体重和平均体重为1克时
综述和结论
本试验中,我们在4种不同的孵化系统中孵化鱼卵,并比较它们的存活率和孵化后早期的生长。到鱼苗平均体重为1克时的存活率整体差异在1%以内,所以没有一种系统有明显优势。
相比立式流水的敞开式底质系统,孵化桶内的鱼苗在开口时的体重要小,到达平均体重为1克是时间要长。与敞开式底质系统相比,其它底质系统没有任何优势,但另外2种不利于清洁和检出死卵。
与立式流水托盘孵化器相比,孵化桶对鱼苗早期增重和早期发育上有负面影响。鱼苗早期的生长对于整体养殖和今后鱼的健康有重要影响。
