Published on Feb. 27, 2020
Incubator hatch comparison
Abstract:
Four different incubation systems commonly used for hatch and yolk sack absorption of salmonid eggs were tested. The systems included a hatching jar, and a vertical flow stack incubator trays arranged in 3 different configurations: open tray, drop in substrate, and built in substrate. Survival, weight at first feeding and average weight at 1g were compared between incubation systems. Survival was not significantly different between systems. Fry hatched in the vertical flow stack incubator open tray configuration were the heaviest at both first feeding and 1g, while those from the hatching jar were on average 14% lighter. Use of substrates in vertical flow stack showed no advantage.
Background
Different types of incubators exist in commercial trout farming. Primarily, vertical flow stacks (Heath stack) incubators, and hatching jar incubators. The function of each is the same, to bring eggs from the eyed stage through hatch and yolk sack absorption, but the hydraulics and environment experienced by hatched fry varies by incubator type. The different environment experience by the fry post hatch could influence stress levels, energy expenditure and overall health. We test this by comparing hatch and ponding parameters of the two different incubator types. One type is the standard upwelling jar, sometimes referred to as a McDonald Upwell. The other type is a vertical stack system, often referred to as a Heath Stack. Within the vertical stack system, we tried three different types of media substrates.
Method
Project Design
Eggs used in this trial were from a standard production lot of the August spawning population. Fertilization and early incubation was done according to Troutlodge standard practices at the Trout Springs facility in Sumner, Washington. Upon reaching approximately 210 accumulated temperature units (ATUs), eggs were processed to remove dead and underdeveloped eggs, then boxed and shipped to the Rock Creek facility in Twin Falls, Idaho.
Upon arrival, eggs were randomly divided into eight batches of 4000 eggs each. Each batch of 4000 was placed into one of the four different incubator types described below. This allowed for two replicates of each incubation device. The eggs were set into the different types of media using a 32oz beaker for transferring. First use spring water (13.2°C) was added to each system at approximately 6 litres per minute. Black Plastic was used to wrap the upwelling jars so that no ambient light would influence the eggs.
Eggs were incubated in the respective systems until yolk sack absorption, at which point they ponded into separate tanks and began eating supplemental feed.
Incubator Systems:
Vertical Flow Stacks
Widely used and commercially available systems that utilize a gravity flow design to incubate eggs. These stacks typically come in 4-8 tray systems but can be stacked on top of one another depending on water quality and flow. They are more often being utilized by state agencies and for research purposes, as they can easily separate small numerations, as well as different types of eggs, in the same system. Often referred to as a Heath Stack.
Substrates for Vertical Flow Incubators
Several different types of substrates are available for the vertical flow incubation systems. The reasoning behind use of a substrate is that by separating the eggs using the media, there will be less spread of fungus, less energy expended from newly hatched alevins, and improved flow throughout the tray. The hatching substrate imitates the gravel substrate naturaly occuring in streams, which provides optimal developmental conditions for fish larvae. The fry are theroretically stronger, healthier, and larger due to increased areas for hiding and less energy expendature.
Vertical Flow Stacks Used:
1. Open Tray Control ( C )
- Even water flow with places for fish to hide away from current
- Water is used in series from top try to bottom tray
- Environment is dark and eggs/fry are protected from light even if lights are turned on.
2. Tray with Substrate Insert ( DI ) :
- Same as above, only with increased area for fish to hide away from current.
- Substrate removeable
3. Tray with Substrate Built In ( BI ) :
- Same as above but media is not removable
Hatching Jar
The hatching jar forces water is to the bottom of the incubator past the developing eggs and out the top of the jar. The advantage to the system is that once the yolk sack is absorbed, fry will flow out the top of the jar to an early rearing tank without any handling. Water flow within the jar, however, can be turbulent, causing fry to utilize energy for swimming rather than growth.
Hatching Jar Used:
Hatching Jar ( J ):
- Upwelling water – provides slightly more turbulent environment
- Single pass water
- Fry are exposed to full light unless ambient light is off.
Project Overview:
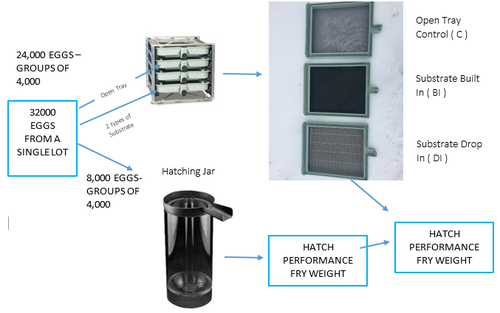
Figure 1: Overall Project Design- showing the different incubations systems used in the trial.
Example of Substrate


Results and discussion
Survival
From the results we can conclude that different incubation medias did not have a significant effect on overall survival. Similar mortality was observed in all systems from eyed egg to hatch, and from hatch to 1g (Table 1). Less than 1% difference in survival was observed across systems, suggesting no clear advantages to any one method. It should be noted that mortality was much easier removed and maintained in the Open Tray Control ( C ) system, compared to all other systems. Staff found both the Built In ( BI ) and Drop In ( DI) substrates to be difficult to use. They did not allow for proper cleaning, fungus developed underneath the substrate in the Drop In system (DI), and flow did not appear to be even throughout the tray.
Length of time between peak hatch and first feeding was similar in all incubation types, averaging around 170 ATU. Peak Hatch defined as more than 75% of the total group having fully hatched.
| Group | Survival to 1g (765.6 ATU) | Survival to 1g (765.6 ATU) |
| Survival % | Deviation from C | |
| C | 88.7% | n/a |
| J | 88.4% | -0.3% |
| BI | 89.3% | 0.7% |
| DI | 89.2% | 0.6% |
Table 1: Survival to 1g average size in the different incubation systems.
Weight
Weight of the fry upon first feeding was 15% less in the jar incubation system, compared to the open tray control (Table 2). We believe this to be the direct result of the amount of energy expelled by fry in the jar system to maintain upright position and combating the required flow inside the jar. Fry hatched in the Built In ( BI ) and Drop In ( DI) substrates were also lighter at first feeding by 2.6% and 1.7% respectively (Table 2, Figure 3), indicating no advantage in energy expenditure over the control.
| Group | First Feeding (171.6 ATU) | First Feeding (171.6 ATU) | 1g (765.6 ATU) | 1g (765.6 ATU) |
| Weight (g)) | Deviation from C | Weight (g) | Deviation from C | |
| C | 0.115 | n/a | 1.02 | n/a |
| J | 0.097 | -15.7% | 0.88 | -13.7% |
| BI | 0.112 | -2.6% | 0.96 | -5.9% |
| DI | 0.113 | -1.7% | 1.01 | -1.0% |
Table 2: ATUs from arrival of eyed eggs to peak hatch, and ATUs from arrival of eyed eggs to 1 gram average.
The same trend was observed at 765 ATU’s when fry in the open tray control reached an average of 1g. At that point, fry in the open tray control were 13.7% heavier than fry hatched in the jar system (Table 2, Figure 3). From this we can conclude that the disadvantage of hatching in the jar incubator extends through to at least 1g, and possibly longer.
Fry hatched in the Built In ( BI ) and Drop In ( DI) substrates were also lighter by 5.9% and 1% respectively, again indicating no advantage over the open tray control (Table 2, Figure 3).
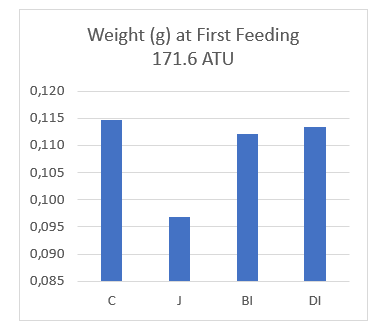
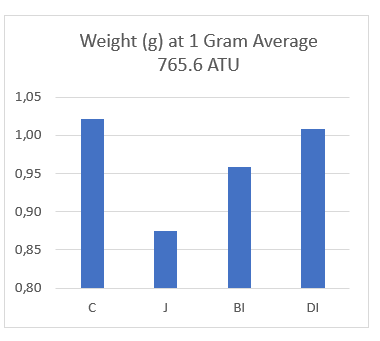
Figure 3: Graphics showing weight of Alevins at first feeding and 1 gram average.
Summary and conclusions
In this trial, we hatched eggs in 4 different incubation systems, and compared survival and early growth of fry hatched in the different systems. Survival to 1g average size was within 1% across all systems, indicating no clear advantage for any one system.
Weight at both first feeding and 1g average size lowest for fry hatched in the hatching jar system, and highest for those hatched in the vertical flow stack open tray control. Neither substrate used in the vertical flow stack showed any advantage over the open tray control, and hindered proper cleaning and removal of dead eggs.
From this trial we can conclude that Hatching Jar incubation method has a negative impact on fry weight and early life stage growth when compared to vertical stack heath trays. This early growth is crucial for the overall success and health of the fish throughout their full life cycle.

